Table of Contents
- Key Features of Quality Management System Software for Pharmaceutical Industry
- Why Quality Management System Software for Pharmaceutical Industry Improves Compliance
- Integration with Other Enterprise Systems
- Choosing the Right Integration Strategy for Quality Management System Software for Pharmaceutical Industry
- Implementation Best Practices
- Tips for Maintaining Long‑Term Success with Quality Management System Software for Pharmaceutical Industry
- Cost Considerations and Return on Investment
- Linking Quality Management with Financial Systems
- Regulatory Landscape and Future Trends
- Preparing for the Next Generation of Quality Management
The pharmaceutical sector operates under a strict regulatory framework that demands meticulous control over every stage of drug development, manufacturing, and distribution. Any lapse in quality can lead to product recalls, legal penalties, and, most importantly, risks to patient safety. Consequently, companies invest heavily in systems that can guarantee consistency, traceability, and compliance.
One of the most powerful tools at their disposal is quality management system software for pharmaceutical industry. This type of software integrates various quality processes—such as document control, deviation handling, change management, and audit tracking—into a single, searchable platform. By automating repetitive tasks and providing real‑time visibility, the software helps organizations meet stringent standards while reducing operational costs.
In the following sections, we will examine the core components of quality management system software for pharmaceutical industry, discuss how it aligns with Good Manufacturing Practices (GMP), and outline best practices for successful deployment.
Key Features of Quality Management System Software for Pharmaceutical Industry
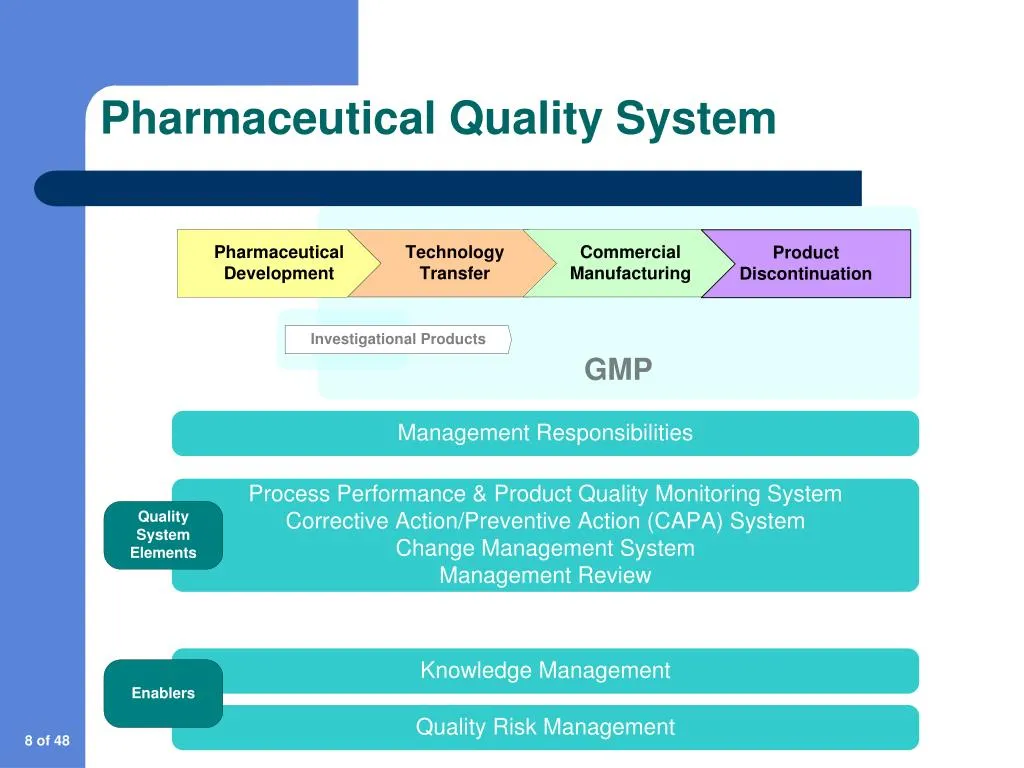
Understanding what makes a quality management system (QMS) effective is essential before selecting a solution. The following capabilities are generally considered non‑negotiable for the pharmaceutical environment.
- Document Control: Centralized repository for SOPs, batch records, and regulatory submissions, with version control and electronic signatures.
- Deviation and CAPA Management: Structured workflows for reporting deviations, investigating root causes, and implementing corrective and preventive actions.
- Change Control: Automated review and approval processes for any modification to processes, equipment, or materials.
- Audit Management: Scheduling, execution, and reporting tools for internal and external audits, ensuring audit trails are complete and searchable.
- Training Management: Tracking of employee qualifications, training records, and competency assessments.
- Risk Management: Integration of Failure Mode Effects Analysis (FMEA) and risk scoring to prioritize actions.
- Electronic Batch Records (EBR): Real‑time capture of manufacturing data, reducing transcription errors and facilitating quick release decisions.
Why Quality Management System Software for Pharmaceutical Industry Improves Compliance
Regulators such as the FDA, EMA, and MHRA expect pharmaceutical companies to demonstrate robust quality controls. Traditional paper‑based systems are prone to gaps, lost documents, and delayed responses. By digitizing these processes, quality management system software for pharmaceutical industry creates immutable audit trails, ensures that only authorized personnel can approve critical changes, and provides instant access to historical data during inspections.
Moreover, the software often includes built‑in validation tools that verify data integrity, enforce electronic signatures compliant with 21 CFR Part 11, and generate reports in formats accepted by regulatory bodies.
Integration with Other Enterprise Systems
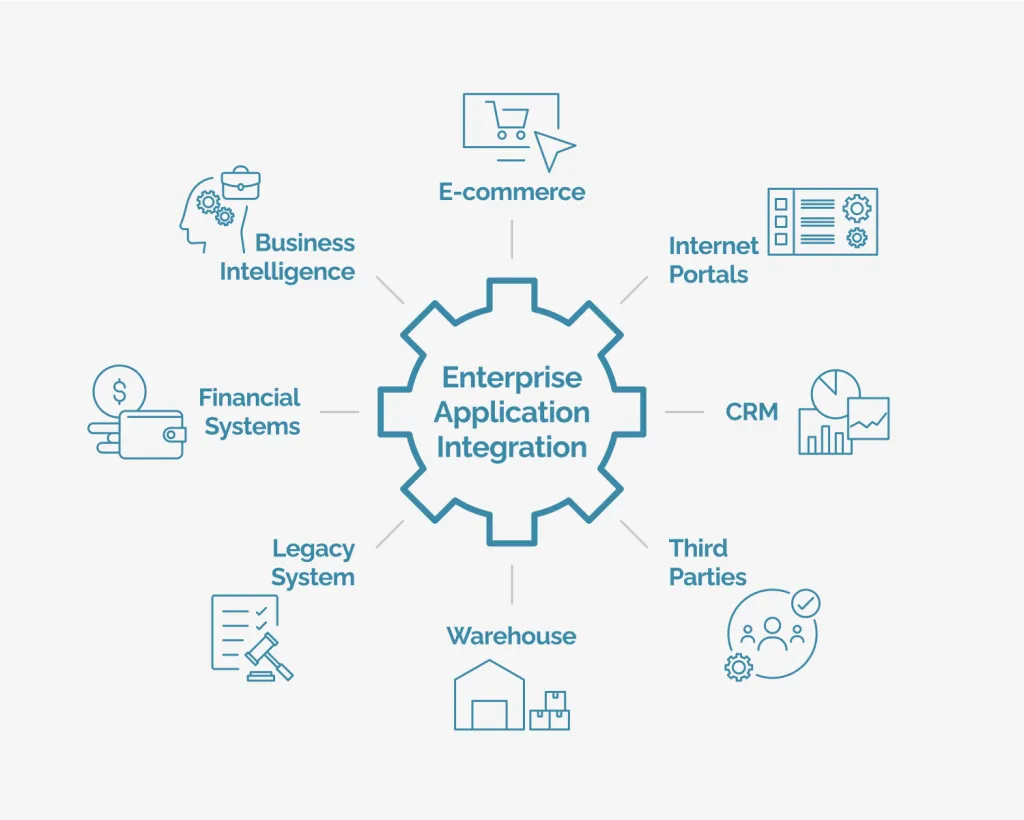
Pharmaceutical operations rarely exist in isolation. To maximize efficiency, quality management system software for pharmaceutical industry must communicate seamlessly with ERP, LIMS, manufacturing execution systems (MES), and even finance platforms. For instance, linking QMS with an ERP can automatically flag non‑conforming inventory, while integration with a LIMS ensures that laboratory test results are instantly reflected in batch release decisions.
Companies looking for a holistic digital ecosystem often evaluate the availability of APIs, pre‑built connectors, and middleware options. When integration is smooth, organizations experience fewer data silos, reduced manual entry, and faster decision‑making.
Choosing the Right Integration Strategy for Quality Management System Software for Pharmaceutical Industry
Start by mapping critical data flows: which systems generate data needed for quality decisions, and where that data must be stored. Next, assess whether the QMS vendor offers certified connectors for your existing ERP or LIMS. If not, consider a third‑party integration platform that supports industry standards such as XML, JSON, or HL7. Finally, validate the integration in a controlled environment before rolling it out to production.
Implementation Best Practices

Deploying quality management system software for pharmaceutical industry is a multi‑phase project that involves people, processes, and technology. Below are proven steps to increase the likelihood of a smooth rollout.
- Executive Sponsorship: Secure commitment from senior leadership to allocate resources and enforce adoption.
- Cross‑Functional Team: Assemble representatives from QA, manufacturing, IT, and regulatory affairs to guide configuration.
- Gap Analysis: Compare current practices against the software’s capabilities to identify required process changes.
- Phased Deployment: Begin with a pilot in one site or product line, gather feedback, and iterate before full‑scale implementation.
- Training and Change Management: Offer hands‑on workshops, e‑learning modules, and continuous support to encourage user confidence.
- Validation and Documentation: Conduct Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ) to meet regulatory expectations.
- Post‑Implementation Review: Monitor key performance indicators (KPIs) such as deviation turnaround time, audit findings, and training compliance.
Tips for Maintaining Long‑Term Success with Quality Management System Software for Pharmaceutical Industry
After go‑live, the system requires ongoing governance. Establish a QMS steering committee that meets quarterly to review system usage, assess new regulatory requirements, and prioritize enhancements. Regularly update SOPs to reflect software upgrades, and conduct refresher training whenever major changes occur.
Another practical tip is to leverage the reporting engine to create dashboards that visualize compliance trends. When management can see real‑time metrics—like the number of open CAPAs or the average audit closure time—they can make proactive decisions that keep the organization ahead of regulatory scrutiny.
Cost Considerations and Return on Investment
The initial outlay for quality management system software for pharmaceutical industry can be significant, encompassing licensing fees, implementation services, and hardware. However, the return on investment (ROI) becomes evident through measurable improvements:
- Reduced labor costs from eliminating manual record‑keeping.
- Faster batch release due to real‑time data access.
- Lower risk of non‑compliance penalties and product recalls.
- Improved supplier quality through integrated vendor management.
Companies often calculate ROI by tracking metrics such as deviation resolution time, audit findings per year, and the cost of regulatory fines avoided. A well‑implemented QMS typically pays for itself within 12‑24 months.
Linking Quality Management with Financial Systems
Financial implications can be further optimized when quality management system software for pharmaceutical industry integrates with accounting solutions. For example, linking deviation costs to the general ledger helps finance teams understand the true cost of quality. An article on Accounts Payable Software Compatible with QuickBooks: A Complete Guide illustrates how such integrations can streamline financial workflows.
Regulatory Landscape and Future Trends
Regulators continue to evolve expectations around data integrity, electronic records, and risk‑based approaches. Quality management system software for pharmaceutical industry must stay current with updates to standards like ICH Q10, EU Annex 11, and FDA’s Digital Manufacturing Initiative.
Emerging technologies—artificial intelligence (AI), machine learning, and blockchain—are beginning to influence QMS capabilities. AI can predict potential deviations by analyzing historical data, while blockchain offers immutable audit trails that satisfy increasing demands for transparency.
Preparing for the Next Generation of Quality Management
Organizations should adopt a modular QMS architecture that allows new functionalities to be added without disrupting existing processes. Engaging with vendors who provide regular feature releases and have a roadmap aligned with regulatory changes ensures long‑term relevance.
Finally, fostering a culture of continuous improvement amplifies the benefits of any software solution. When employees view quality as a shared responsibility rather than a compliance checkbox, the entire organization becomes more resilient to both operational and regulatory challenges.
In summary, quality management system software for pharmaceutical industry is not merely an IT investment; it is a strategic enabler that safeguards product integrity, enhances operational efficiency, and positions companies for future growth. By selecting a solution that offers robust core functionalities, seamless integration, and a clear implementation pathway, pharmaceutical manufacturers can meet today’s compliance demands while preparing for tomorrow’s innovations.